Forms of Energy
There are several types of energy, for Biology 12 we will look at:
Kinetic energy - the energy associated with motion; this is the energy that objects have when they're moving.
Potential energy - the energy from position; when potential energy is stored for chemical reactions, we call it chemical energy.
Thermal energy - energy of molecular movement; the warmer something is, the greater its thermal energy.
Light energy - energy from the sun; plants use light energy for photosynthesis. This is the ultimate source of energy for almost all organisms on Earth.
To give you a good idea what each kind of energy is, think about this scenario. Light energy is used to grow the food you eat. Some of that energy is then stored as chemical energy within your muscles. When you use your arms to lift a ball into the air, you are transforming some chemical energy into kinetic energy for muscle movement. By raising the ball, your are also transforming your kinetic energy into the ball's potential energy. When you release the ball, the potential energy o the ball is converted into kinetic energy and the ball drops to the ground. When it hits the ground, some kinetic energy is dissipated into heat energy, which slightly warms up the ground and itself. The rest of the kinetic energy of the ball will be re-converted into potential energy as the ball bounces back into the air. This cycle continues until all of the ball's energy has been dissipated into heat energy or lost to air friction.
We usually measure energy in joules and kilojoules, but sometimes we use calories and kilocalories instead. It is important to know how to convert these values. 1 calorie is the same as 4.18 joules, and 1 joule is equal to 0.239 calories.
Kinetic energy - the energy associated with motion; this is the energy that objects have when they're moving.
Potential energy - the energy from position; when potential energy is stored for chemical reactions, we call it chemical energy.
Thermal energy - energy of molecular movement; the warmer something is, the greater its thermal energy.
Light energy - energy from the sun; plants use light energy for photosynthesis. This is the ultimate source of energy for almost all organisms on Earth.
To give you a good idea what each kind of energy is, think about this scenario. Light energy is used to grow the food you eat. Some of that energy is then stored as chemical energy within your muscles. When you use your arms to lift a ball into the air, you are transforming some chemical energy into kinetic energy for muscle movement. By raising the ball, your are also transforming your kinetic energy into the ball's potential energy. When you release the ball, the potential energy o the ball is converted into kinetic energy and the ball drops to the ground. When it hits the ground, some kinetic energy is dissipated into heat energy, which slightly warms up the ground and itself. The rest of the kinetic energy of the ball will be re-converted into potential energy as the ball bounces back into the air. This cycle continues until all of the ball's energy has been dissipated into heat energy or lost to air friction.
We usually measure energy in joules and kilojoules, but sometimes we use calories and kilocalories instead. It is important to know how to convert these values. 1 calorie is the same as 4.18 joules, and 1 joule is equal to 0.239 calories.
Metabolic Pathways:
Simply put, a metabolic pathway is a model of the steps necessary for a reactant molecule to become product molecule(s). It also includes the enzymes necessary for each step of the pathway. A metabolic pathway may look something like this:
A ----------------> B ----------------> C ---------------> D
enzyme 1 enzyme 2 enzyme 3
Some metabolic pathways break down complex molecules into simpler products, releasing energy in the process. These pathways are called catabolic pathways. In contrast, pathways that consume energy to form complex molecules from simpler reactants are called anabolic pathways.
A ----------------> B ----------------> C ---------------> D
enzyme 1 enzyme 2 enzyme 3
Some metabolic pathways break down complex molecules into simpler products, releasing energy in the process. These pathways are called catabolic pathways. In contrast, pathways that consume energy to form complex molecules from simpler reactants are called anabolic pathways.
Laws of Thermodynamics
Thermodynamics refer to the energy transformations which occur within systems. There are two types of systems, closed and open systems. In an open system, energy and matter can be transferred to and from its surroundings. In a closed system, energy and matter cannot be transferred to or from the outside. An example of an open system could be an animal, which absorbs its energy from consuming external sources of food, and releases heat energy and wastes into its surroundings. There are no perfectly closed systems, because eventually energy in a system will find a way to escape. However, we can say a thermos bottle is a nearly closed system because it only allows a very slow rate of energy transfer in and out of the system. The only perfectly closed system is the universe.
There are two laws which govern the transformation and transfer of energy within systems, the first and second laws of thermodynamics.
The first law of thermodynamics states that the total amount of energy in the universe remains constant. Therefore, energy can be transferred and transformed, but it cannot be created or destroyed. When you bounce a ball, it appears that energy is being lost as the ball eventually comes to a rest, but that is because when the ball hits the ground, some of its energy is converted into heat energy. Likewise, when the ball moves through the air, air friction slows down the ball and converts some of its energy into heat. But that's not all, when the ball bounces, it releases sound energy. These are all ways in which a system can dissipate energy. So energy is always conserved, even though a system may lose its energy to the outside environment.
The second law of thermodynamics is a bit more complex. It states that every energy transfer or transformation increases the disorder of the universe. We use entropy to express disorder, so the more entropy present in a system, the greater the disorder. Note that organisms are considered to be highly organized systems. However, this does not violate the second law because organisms are also creating more entropy for the universe through dissipating heat. This is more than enough compensation for the decrease in entropy associated with their growth.
When we call a reaction spontaneous, we mean that it can occur without a net input of energy from the outside. The only way this can happen is if it increases the entropy of the universe in the process. Contrary to the spontaneous's literary connotation, spontaneous events can occur slowly or rapidly.
There are two laws which govern the transformation and transfer of energy within systems, the first and second laws of thermodynamics.
The first law of thermodynamics states that the total amount of energy in the universe remains constant. Therefore, energy can be transferred and transformed, but it cannot be created or destroyed. When you bounce a ball, it appears that energy is being lost as the ball eventually comes to a rest, but that is because when the ball hits the ground, some of its energy is converted into heat energy. Likewise, when the ball moves through the air, air friction slows down the ball and converts some of its energy into heat. But that's not all, when the ball bounces, it releases sound energy. These are all ways in which a system can dissipate energy. So energy is always conserved, even though a system may lose its energy to the outside environment.
The second law of thermodynamics is a bit more complex. It states that every energy transfer or transformation increases the disorder of the universe. We use entropy to express disorder, so the more entropy present in a system, the greater the disorder. Note that organisms are considered to be highly organized systems. However, this does not violate the second law because organisms are also creating more entropy for the universe through dissipating heat. This is more than enough compensation for the decrease in entropy associated with their growth.
When we call a reaction spontaneous, we mean that it can occur without a net input of energy from the outside. The only way this can happen is if it increases the entropy of the universe in the process. Contrary to the spontaneous's literary connotation, spontaneous events can occur slowly or rapidly.
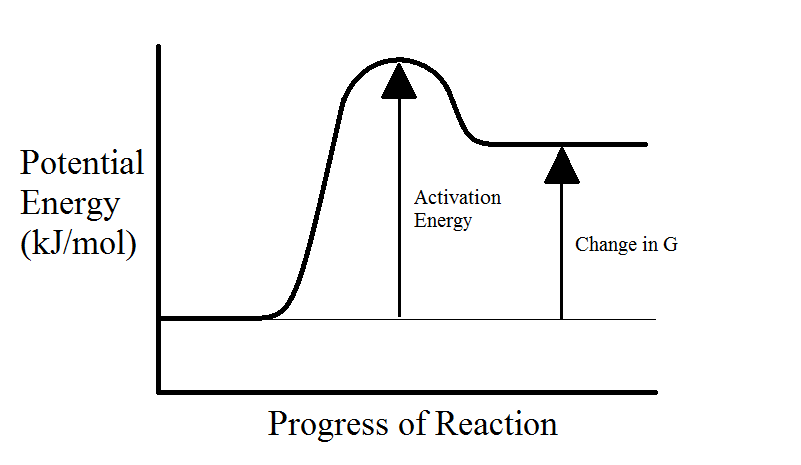
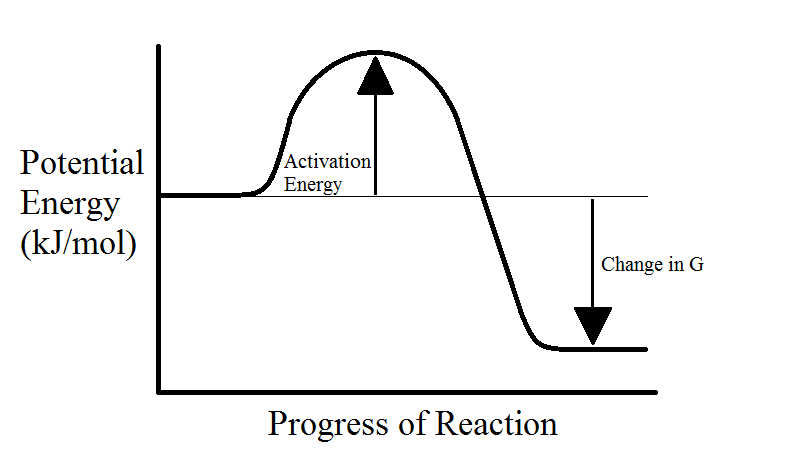
Endergonic vs. Exergonic Reactions
When a chemical reactions happens within an organism, energy is either consumed or released. If the reactants consume energy to make higher-energy products, then the reaction is endergonic. On the other hand, if the reactions release energy to form lower-energy products, then the reaction is exergonic. Catabolic reactions are exergonic because they release energy, and anabolic reactions are endergonic because they consume energy.
Even though exergonic reactions are spontaneous, they do not always happen quickly. This is because reactants in either exergonic or endergonic reactions must overcome an activation energy barrier before anything occurs. This is demonstrated in the following diagrams.
Even though exergonic reactions are spontaneous, they do not always happen quickly. This is because reactants in either exergonic or endergonic reactions must overcome an activation energy barrier before anything occurs. This is demonstrated in the following diagrams.
Often reactants do not have enough energy to overcome the activation energy barrier, this is the main reason why high-energy molecules can exist in large numbers. In the body, we have enzymes which selectively facilitate the reactions by lowering the activation energy barrier (see next section).