Enzymes
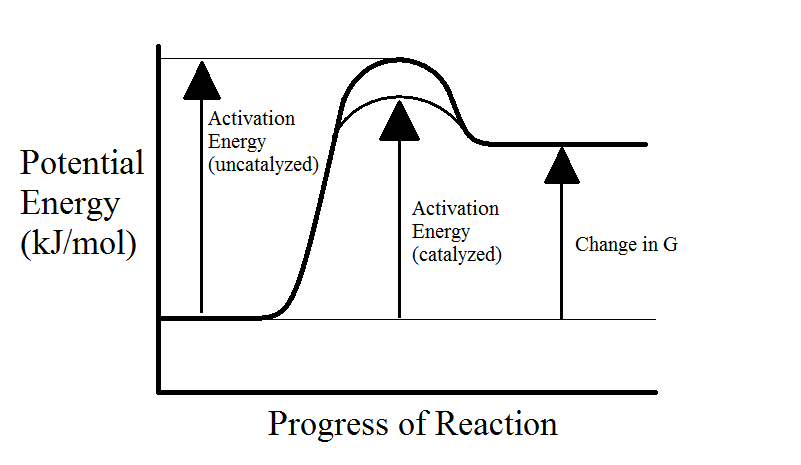
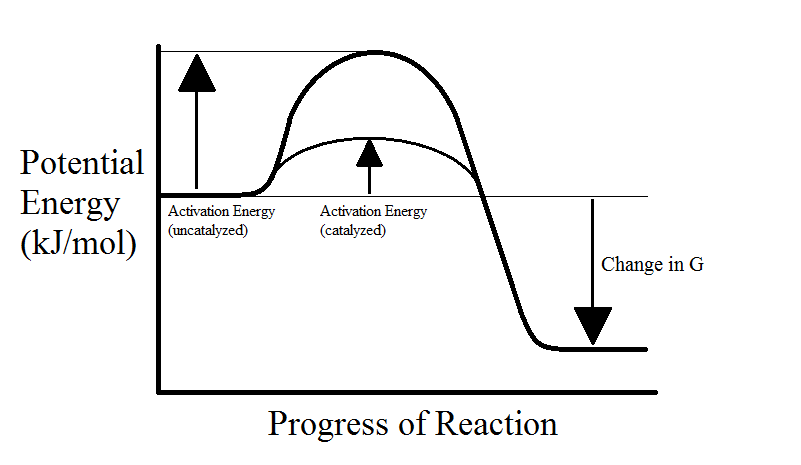
Enzymes are proteins that speed up chemical reactions. As we learned earlier, the activation energy barrier makes it more difficult for reactants to form products. Enzymes facilitate reactions by lowering the activation energy. This is accomplished through the following methods:
1. Providing a template for the substrates to come together in the correct orientation
2. Stretching the substrates towards their transition state conformation and pressuring the bonds which must be broken during the reaction
3. Providing a more suitable microenvironment for the reaction to occur (e.g. temperature and pH)
4. Temporary covalent bonding of the enzyme's R groups to the substrates
Note that enzymes do not have any effect on the energy of the products or the equilibrium of the reaction. They're only capable of changing the reaction rate. In addition, even if an enzyme is present, some reactants will still take the uncatalyzed pathway with higher activation energy (see below graphs).
1. Providing a template for the substrates to come together in the correct orientation
2. Stretching the substrates towards their transition state conformation and pressuring the bonds which must be broken during the reaction
3. Providing a more suitable microenvironment for the reaction to occur (e.g. temperature and pH)
4. Temporary covalent bonding of the enzyme's R groups to the substrates
Note that enzymes do not have any effect on the energy of the products or the equilibrium of the reaction. They're only capable of changing the reaction rate. In addition, even if an enzyme is present, some reactants will still take the uncatalyzed pathway with higher activation energy (see below graphs).
Enzyme Structure
The enzyme's structure is closely related to its function. They selectively bond to certain substrates (reactants) and speed up the reaction between them. This process occurs only at the active site of the enzyme. The active site is usually a small pocket on the surface of the enzyme. It can change its shape slightly in order to better accommodate the substrates, this is called induced fit. When the enzyme and the substrates bond, they form an enzyme-substrate complex, which is basically a temporary combination until the substrates turn into products and are released.
Many enzymes need cofactors in order to function properly. Cofactors are attachments to the enzyme which enhance its function. If a cofactor is non-organic (like metal ions), we simply call it a cofactor, but if a cofactor is organic (like vitamins), then we can call it a coenzyme.
Many enzymes need cofactors in order to function properly. Cofactors are attachments to the enzyme which enhance its function. If a cofactor is non-organic (like metal ions), we simply call it a cofactor, but if a cofactor is organic (like vitamins), then we can call it a coenzyme.
Effect of Surroundings on Enzymes
An enzyme's activity depends on environmental factors. This is because at extreme temperatures or pH, the tertiary structure of the enzyme begins to fall apart (denature) and therefore it can no longer function. Due to their sensitivity to the environment, enzymes have an optimal temperature and pH in which it functions best. For humans, most of our enzymes function best at temperatures of around 35-40 degrees Celsius. However, for extremophile bacteria living in very hot conditions, their enzymes may have an optimal temperature of over 70 degrees Celsius. Just like how enzymes have an optimal temperature, they also have an optimal pH. Most of our enzymes work best at pH's of around 7. However, pepsin, an enzyme found in the acidic conditions of our stomach, has an optimal pH of about 2, whereas trypsin, an intestinal enzyme which works in alkaline conditions, has an optimal pH of 8.
In order to maximize enzymatic efficiency in a cell, one would have to alter the cellular conditions so the temperature and pH are within the optimal range for the enzymes. In our cells, the cellular conditions are usually kept at optimal levels. A cell that suddenly experiences a cytosol pH change from 7 to 4 would mostly likely die because their enzymes denature from the acidity. Likewise, the reason why getting a high fever is dangerous is because our enzymes start to denature at temperatures higher than 40 degrees Celsius.
In order to maximize enzymatic efficiency in a cell, one would have to alter the cellular conditions so the temperature and pH are within the optimal range for the enzymes. In our cells, the cellular conditions are usually kept at optimal levels. A cell that suddenly experiences a cytosol pH change from 7 to 4 would mostly likely die because their enzymes denature from the acidity. Likewise, the reason why getting a high fever is dangerous is because our enzymes start to denature at temperatures higher than 40 degrees Celsius.
Other Factors that Affect Enzymes
|
If the substrate concentration increases, the rate of reaction increases as well. This is because free enzymes will find it easier to bind to new substrates if there are more of them. However, when the substrate concentration is high enough, the enzymes will be constantly bonded to them, and from that point on, increasing the substrate concentration will have no more effect on the rate of reaction. At this point, we consider the enzymes saturated because they are working 100% of the time.
If the enzyme concentration increases, the rate of reaction increases proportionally as well. If the competitive inhibitor concentration increases, the rate of reaction decreases. This is because there is a smaller chance for the enzymes to attach to the substrate, and a greater chance of them binding to the inhibitors instead. If the non-competitive inhibitor concentration increases, the rate of reaction decreases. This is because there are less enzymes that still have its active site in the regular conformation. Note that no matter how much both competitive and/or non-competitive inhibitors are present, one can never have a negative reaction rate. That is, even if all the enzymes are inhibited, they cannot reverse the reaction by converting the products to reactants. If the product concentration is increased, they may act as competitive inhibitors or trigger the release of competitive inhibitors, thereby lowering reaction rate. The relative location of enzymes in the cell also affect their activity. Enzymes that work together in a specific metabolic pathway tend to stick close together so one enzyme can quickly pass its product molecules to the next enzyme as its substrates. |